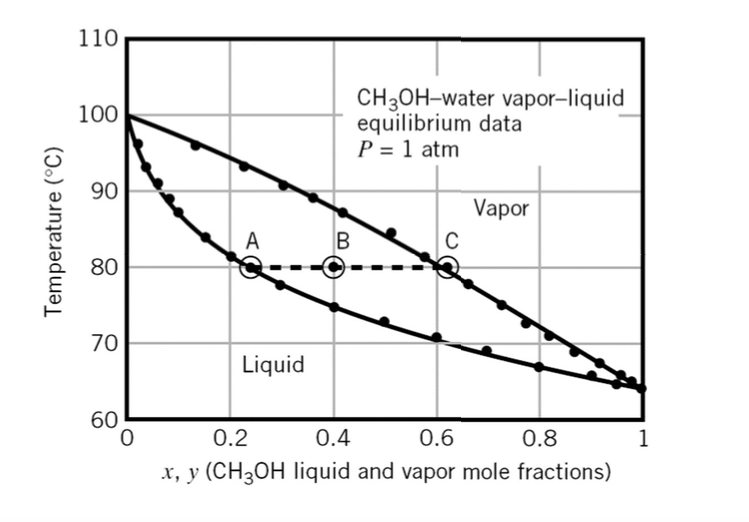
For vapor-liquid equilibrium data of a methanol-water mixture, What is the K value of methanol at a methanol liquid mole fraction of 0.2? | Homework.Study.com

Isobaric Vapor–Liquid Equilibrium for Methanol + Dimethyl Carbonate + 1-Octyl-3-methylimidazolium Tetrafluoroborate | Journal of Chemical & Engineering Data
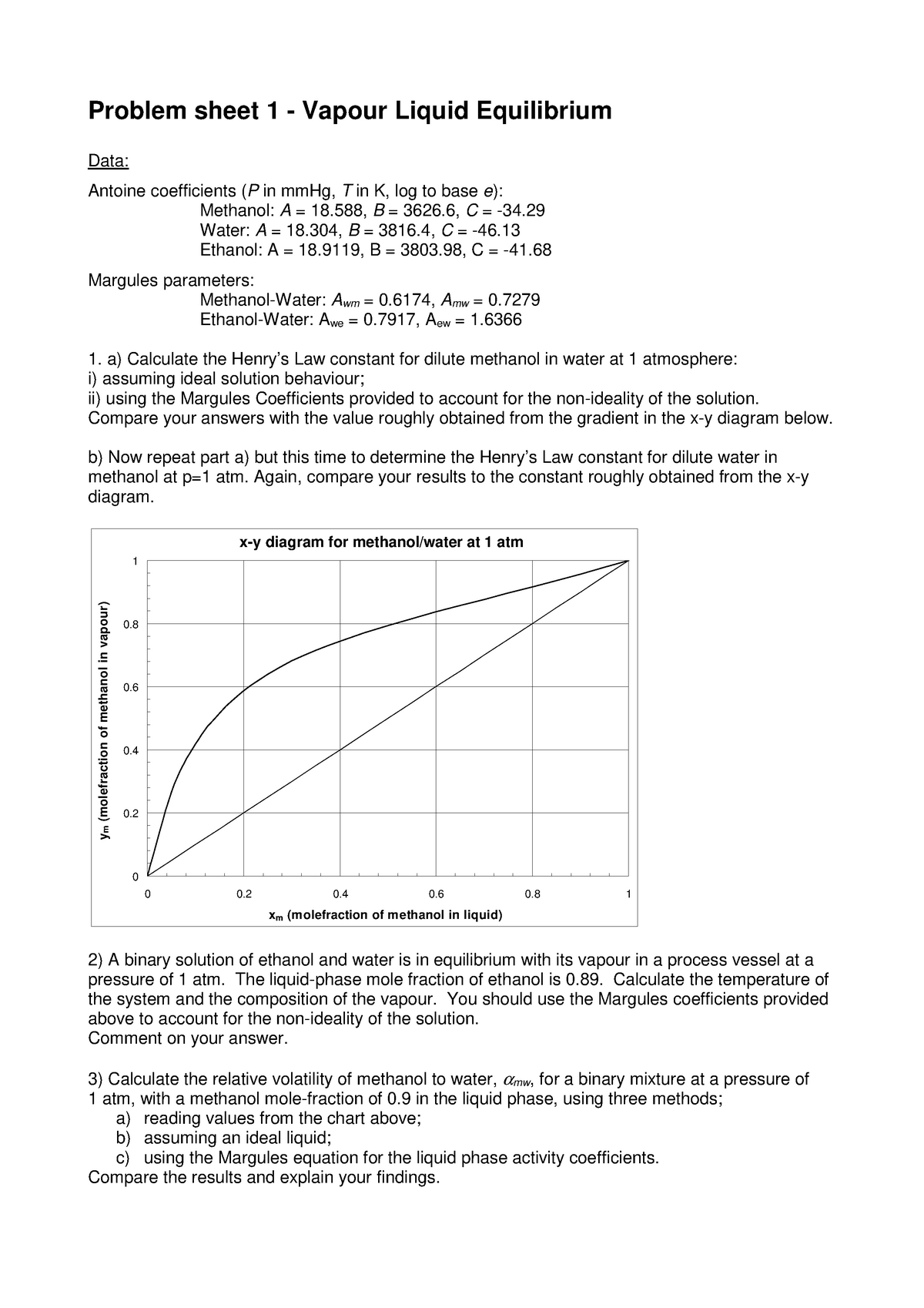
Problem sheet 1 - Water: A = 18, B = 3816, C = -46. Ethanol: A = 18, B = 3803, C = -41. Margules - Studocu
![PDF] Effect of Inorganic Salts on the Isobaric Vapor–Liquid Equilibrium of the Ethyl Acetate–Ethanol System | Semantic Scholar PDF] Effect of Inorganic Salts on the Isobaric Vapor–Liquid Equilibrium of the Ethyl Acetate–Ethanol System | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/1c511fcf522437a07d479f5f6ddaa2c74aba0705/6-Table9-1.png)
PDF] Effect of Inorganic Salts on the Isobaric Vapor–Liquid Equilibrium of the Ethyl Acetate–Ethanol System | Semantic Scholar

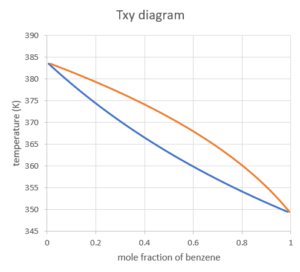
T - x 1 -y1 diagram for methanol (1) + water (2) at 101.325 kPa: □,... | Download Scientific Diagram

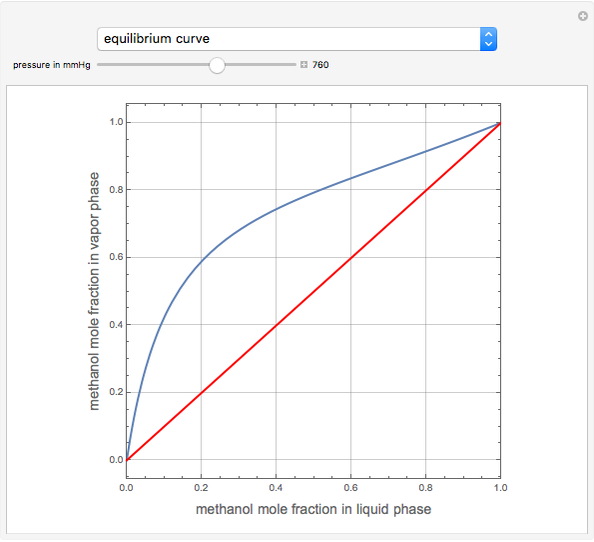
T-x-y and x-y Diagrams for Binary Vapor-Liquid Equilibrium (VLE) in Flash Drum - Wolfram Demonstrations Project
![Vapor–Liquid Equilibrium of Ionic Liquid 7-Methyl-1,5,7-triazabicyclo[4.4.0]dec-5-enium Acetate and Its Mixtures with Water | Journal of Chemical & Engineering Data Vapor–Liquid Equilibrium of Ionic Liquid 7-Methyl-1,5,7-triazabicyclo[4.4.0]dec-5-enium Acetate and Its Mixtures with Water | Journal of Chemical & Engineering Data](https://pubs.acs.org/cms/10.1021/acs.jced.9b01039/asset/images/acs.jced.9b01039.social.jpeg_v03)
Vapor–Liquid Equilibrium of Ionic Liquid 7-Methyl-1,5,7-triazabicyclo[4.4.0]dec-5-enium Acetate and Its Mixtures with Water | Journal of Chemical & Engineering Data
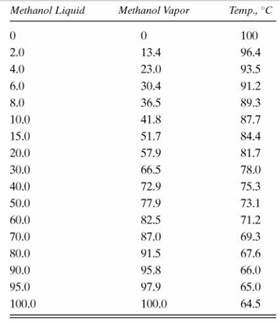
Solved) - Two flash distillation chambers are hooked together as shown in... - (1 Answer) | Transtutors

Processes | Free Full-Text | Thermodynamics and Machine Learning Based Approaches for Vapor–Liquid–Liquid Phase Equilibria in n-Octane/Water, as a Naphtha–Water Surrogate in Water Blends

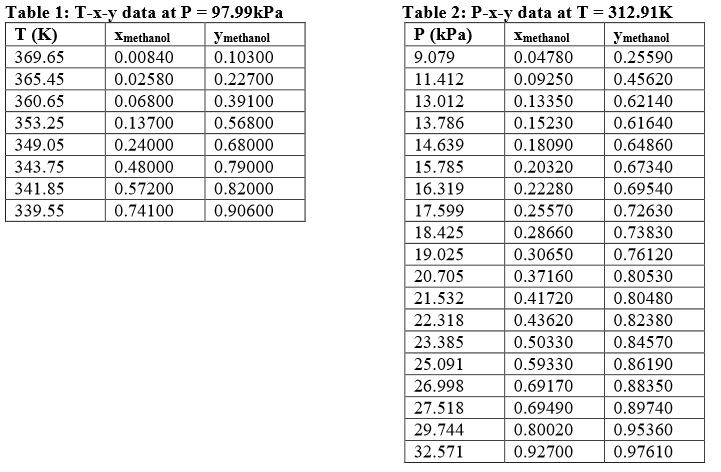

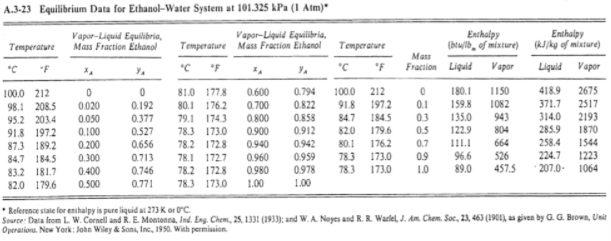



![Solved] A methanol?water feed stream is introduce | SolutionInn Solved] A methanol?water feed stream is introduce | SolutionInn](https://s3.amazonaws.com/si.experts.images/questions/2022/11/636a40e8ef34d_816636a40e8e139a.jpg)